Highly specific, and selective for SARS-CoV-2 nucleocapsid protein
Simple, visual results within 15 minutes
Specimen Type: Direct Nasopharyngeal Swab*
No Instrumentation Required
Storage: 2-30°C
Stability: 2 years from date of manufacture
Controls available separately
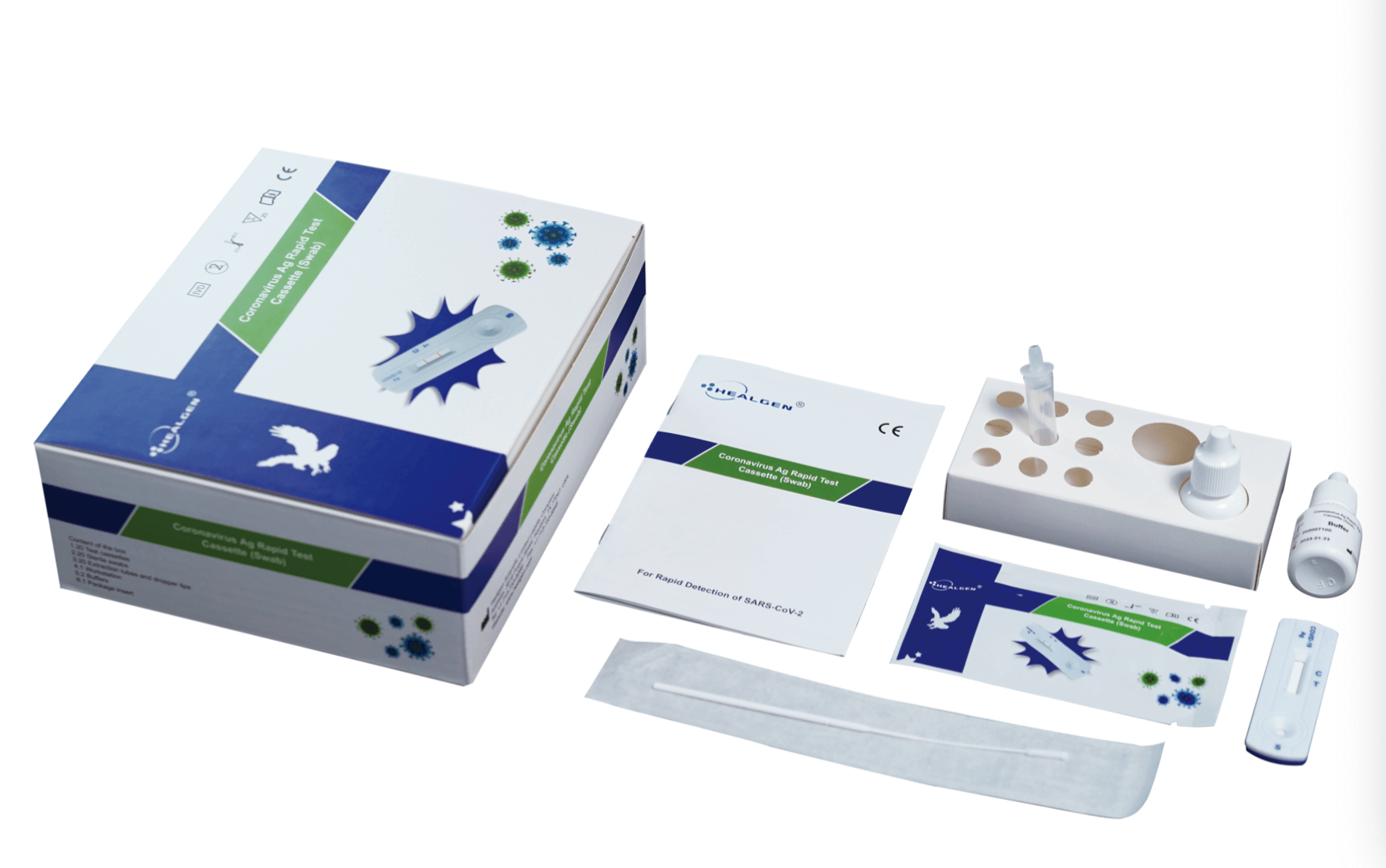
Rapid Covid-19 Antigen Test
The Rapid COVID-19 Antigen Test is an in vitro immunochromatographic assay for the qualitative detection of nucleocapsid protein antigen from SARS-CoV-2 through direct nasopharyngeal (NP) swab specimens directly collected from individuals who are suspected of having COVID-19 by their healthcare professional. This test is designed to assist in the rapid diagnosis of SARS-CoV-2 infections. The Rapid COVID-19 Antigen Test does not determine between SARS-CoV and SARS-CoV-2.
For questions or pricing information please call us at (866) 788-2855
This test has not been reviewed by the FDA
Negative results do not rule out SARS-CoV-2 infection, particularly those who have been in contact with the virus. Follow up testing with a molecular diagnostic test should be considered to rule out infection in these individuals.
Use of this test is limited to laboratories certified to perform high complexity testing, including testing at point-of-care when the site is covered by the laboratory's CLIA certificate of high-complexity testing.
This test is not for home-use or at-home specimen collection.