An infectious control plan is no longer optional for healthcare-adjacent workplaces in 2026. Healthcare facilities, skilled nursing facilities, residential care communities, and large employers must maintain clear policies and procedures for infection control to reduce outbreaks, maintain staffing levels, and remain compliant with federal and state guidance. Below, you’ll find a structured framework for developing an infectious control plan, including infectious disease preparedness, COVID workplace testing policy development, and screening best practices tailored for high-risk environments.
Quick Summary
- Key Components of an Effective COVID & Flu Infectious Control Plan
- Building An Infectious Control Plan: Step-By-Step Preparation
- Test Screening
- Maintaining And Updating Your Flu & COVID-19 Infectious Control Plan In 2026
Key Components of an Effective COVID & Flu Infectious Control Plan
An effective infectious control plan outlines how your organization prevents, identifies, and responds to communicable illnesses such as COVID-19 and seasonal influenza. For healthcare facilities and high-density workplaces, infection control policy and procedures must be documented, communicated, and consistently enforced.
Core components typically include:
- Written policies and procedures of infection control
- Employee illness reporting protocols
- Isolation and return-to-work criteria
- Environmental cleaning standards
- PPE usage requirements
- Testing and screening guidelines
- Communication plans for staff
In nursing homes and rehabilitation facilities, regulatory agencies often expect written documentation that can be reviewed during inspections or audits. A well-structured infectious control plan also demonstrates proactive infectious disease preparedness, reducing liability exposure and operational disruption.
How Does Infectious Disease Preparedness Protect Staffing Levels?
Infectious disease preparedness directly affects workforce stability. When outbreaks spread unchecked, facilities face absenteeism, overtime strain, and service interruptions. A structured infectious control plan helps identify cases early, isolate appropriately, and limit transmission.
Testing policies, symptom screening, and rapid response procedures reduce the number of employees sidelined at any given time. For staffing agencies and healthcare administrators, this stability protects patient care standards and revenue continuity. Planning ahead also prevents reactive decisions that may lead to inconsistent enforcement or confusion among employees.
Building An Infectious Control Plan: Step-By-Step Preparation
The following framework helps ensure your infection control policy and procedures address compliance, staffing, and operational needs.
Step 1: Conduct A Risk Assessment
Begin by evaluating your workplace risk factors:
- Population served (elderly, immunocompromised, general workforce)
- Facility layout and ventilation
- Shared spaces and high-contact surfaces
- Staffing density and shift overlap
- History of outbreaks
Healthcare and long-term care settings typically require higher-level precautions due to vulnerable residents. Documenting these findings establishes the foundation of your infectious control plan and supports regulatory defensibility.
Step 2: Develop Written Policies And Procedures Of Infection Control
Your written policies and procedures of infection control should clearly define:
- Illness reporting requirements
- Isolation protocols
- Return-to-work standards
- PPE guidelines
- Visitor policies
- Cleaning and disinfection schedules
Written documentation ensures consistency. Supervisors and HR personnel must understand how to apply these policies fairly and uniformly. Written policies also reduce confusion during active outbreaks.
Step 3: Create A Flu And COVID Workplace Testing Policy
A structured flu and COVID workplace testing policy is central to infectious disease preparedness in 2026. Testing policies should specify:
- Who is tested (symptomatic staff, exposed staff, routine screening groups)
- Testing frequency
- Type of test used (antigen vs. molecular)
- Documentation and recordkeeping standards
- Confidentiality safeguards
Facilities that rely on rapid testing can identify cases within minutes, limiting exposure and accelerating response time.
Step 4: Train Staff
Training ensures your COVID and flu infectious control plan functions properly. Employees should understand:
- Symptoms of COVID-19 and influenza
- When to report illness
- Proper PPE use
- Testing procedures
- Cleaning expectations
Training sessions should be documented and refreshed annually or when guidance changes. In healthcare settings, competency validation may be required.
Step 5: Stock Adequate Testing Supplies
An infectious control plan cannot function without sufficient testing inventory. Facilities should maintain a consistent supply of rapid COVID and flu tests to avoid shortages during peak seasons.
Test Screening
Testing plays a central role in infection control policy and procedures. Screening protocols should align with risk level, regulatory requirements, and staffing capacity.
COVID-19 And Flu Test Types
Two primary categories are used in workplace settings:
Rapid Antigen Tests
- Provide results in 10–15 minutes
- Ideal for routine screening
- Cost-effective for bulk purchasing
Molecular (PCR) Tests
- Higher sensitivity
- Laboratory processing required
- Longer turnaround time
Many facilities use antigen tests for routine screening and reserve PCR tests for confirmatory purposes. For high-risk environments, consistent access to bulk rapid testing supports infectious disease preparedness goals.
Budgeting and Cost Planning for Workplace Testing
Cost planning is essential for long-term sustainability. Consider:
- Per-test pricing
- Frequency of testing
- Seasonal surges
- Storage requirements
- Staff time for administration
Bulk purchasing significantly reduces per-unit costs. Organizations that maintain structured screening schedules can forecast expenses more accurately.
Partnering with a supplier that specializes in bulk drug and infectious disease testing solutions helps facilities manage both compliance and cost control. Drug Tests In Bulk supplies professional-use rapid testing kits in bulk quantities designed for workplace and healthcare settings.
Recommended Products
For organizations building an infectious control plan in 2026, DrugTestsInBulk (DTIB) recommends:
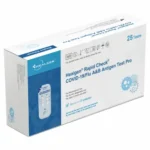
HEALGEN RAPID COVID-19 / FLU A&B ANTIGEN TEST CASSETTE (SWAB)
The Healgen Rapid COVID-19 / Flu A&B Antigen Test Cassette provides dual detection of COVID-19 and Influenza A & B using a single nasal swab.
Key Features:
- FDA EUA authorized
- 20 tests per case
- Clear visual readout
- Quick turnaround results
- Suitable for professional use
- Efficient screening during the respiratory illness season
These disposable face masks offer a comfortable fit for daily use and provide practical protection in the workplace and healthcare environments.
Key Features:
- Elastic Ear Loops
- 175mm X 95mm (Medium)
- 1,000 masks per order
Drug Tests In Bulk offers free shipping and bulk ordering options tailored for employers, rehabilitation centers, and healthcare facilities that require reliable inventory during the respiratory illness season.
Maintaining And Updating Your Flu & COVID-19 Infectious Control Plan In 2026
An infectious control plan must remain current. Public health recommendations continue to evolve, especially regarding testing frequency and isolation timelines.
Review your infection control policy and procedures:
- Annually
- After regulatory updates
- Following the outbreak events
- When introducing new testing technology
Document each revision and communicate changes clearly to staff. Routine reviews strengthen infectious disease preparedness and demonstrate proactive compliance during inspections.
To sum up, a well-developed COVID-19 and flu infectious control plan protects patients, employees, and operational continuity. Nursing homes, rehabilitation centers, and workplace environments must implement written infectious control policies and procedures supported by structured testing and training. With reliable access to rapid testing supplies and clear COVID workplace testing policy guidelines, organizations can respond quickly to illness threats while protecting staffing levels and regulatory standing.
For facilities seeking dependable bulk testing solutions, Drug Tests In Bulk offers professional-grade rapid COVID and flu tests designed for workplace screening programs.
Frequently Asked Questions About Building A Workplace COVID-19 and Flu Infectious Control Plan In 2026
Who Needs An Infectious Control Plan?
Healthcare facilities, nursing homes, rehabilitation centers, staffing agencies, and large employers benefit from maintaining an infectious control plan. High-density workplaces and organizations serving vulnerable populations face the greatest need for structured infectious disease preparedness.
How Often Should An Infectious Control Plan Be Updated?
At a minimum, review your COVID and flu infectious control plan annually. Additional updates should occur when public health guidance changes or after an outbreak exposes procedural gaps.
What Types Of Tests Are Used In An Infectious Control Plan?
Most facilities rely on rapid infectious disease antigen tests for routine screening and molecular tests for confirmation. Combo COVID and flu antigen tests are increasingly common for respiratory season screening, with professional-grade options like the Healgen Rapid COVID-19 / Flu A&B Antigen Test Cassette (Swab) offered by Drug Tests In Bulk (DTIB) for institutional purchasing.
Is An Infectious Control Plan Required By Law?
Requirements vary by state and industry. Healthcare facilities and long-term care providers often face regulatory mandates requiring the maintenance of written infection control policies and procedures. Employers should review federal, state, and local guidance to ensure compliance.